Saving the Environment and Bottom Lines by Reducing Emissions



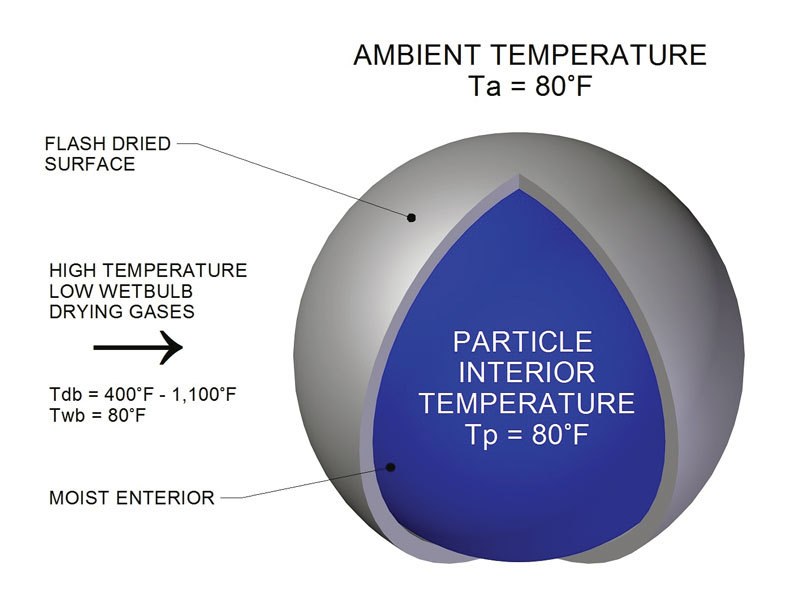
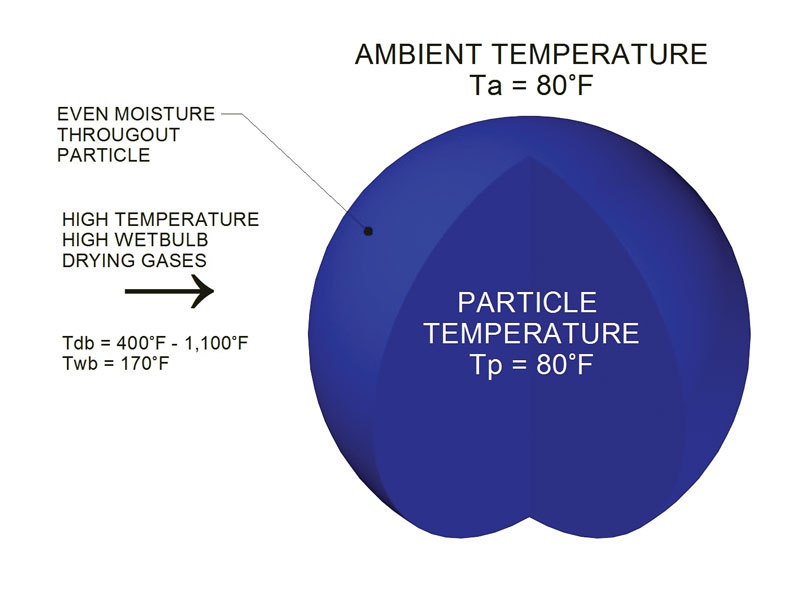
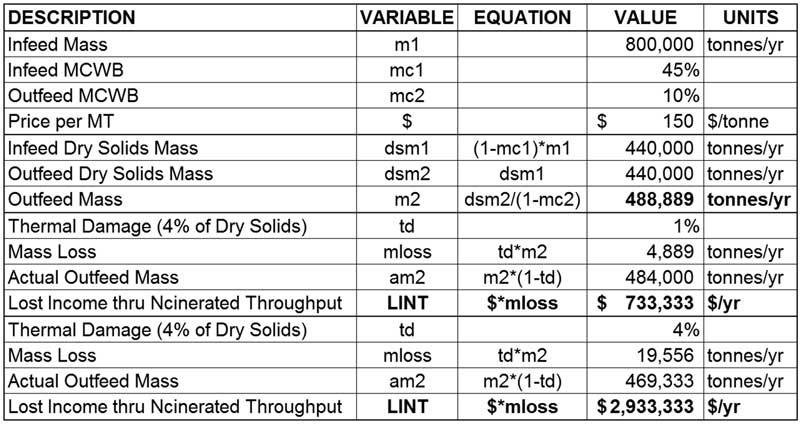
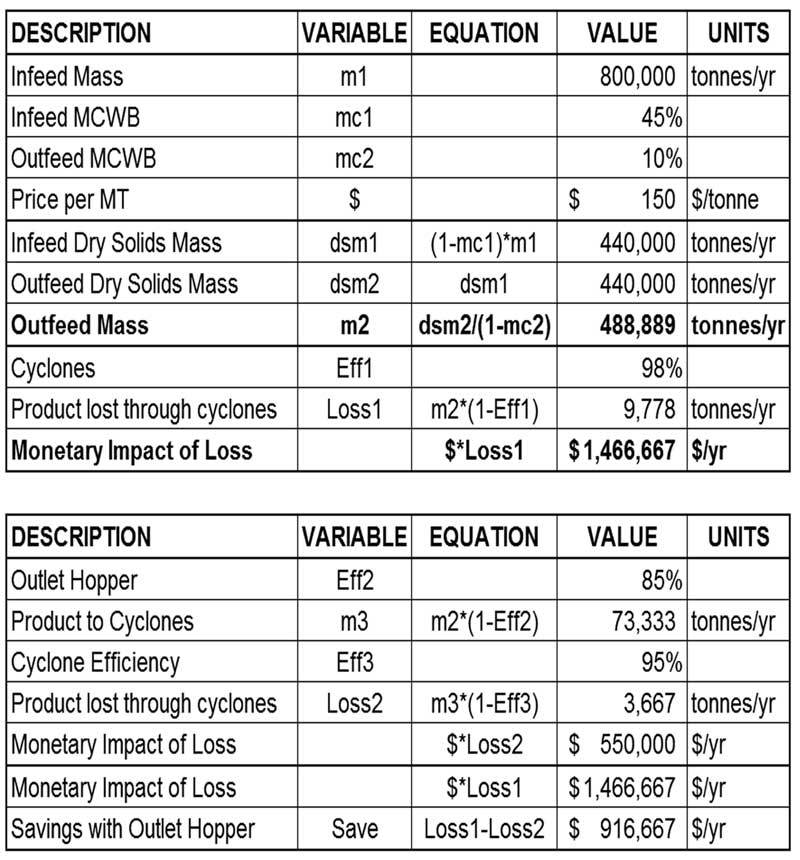









PHOTO: THOMPSON DRYERS
May 20, 2019
BY Becky Long
Advertisement
Advertisement
Related Stories
The USDA on April 23 awarded more than $194 million in loans and grants through the agency’s Rural Energy for America Program. Biogas and bioenergy projects were among those selected to receive funding.
Drax Group on April 18 announced a carbon removals deal with C-Zero Markets, an environmental consultancy. Under the deal, C-Zero will purchase carbon dioxide removals credits from Drax representing 2,000 metric tons of permanently stored carbon.
A bill introduced by Sen. Cory Booker, D-N.J., would require the U.S. EPA to alter the way it assesses lifecycle GHG emissions from forest biomass. ABEA stresses that any accurate assessment must account for the alternative fate of biomass fuels.
Moisture in wood and biomass operations impacts product as well as equipment, energy usage, production efficiency, downtime and more.
A bioenergy with carbon capture and storage (BECCS) project under development in Sweden by Stockholm Energi was awarded planning approval on March 28 by the country’s Land and Environmental Court.





